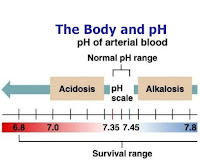
A respiratory acidosis is a primary acid-base disorder in which arterial pCO2 rises to a level higher than expected.
At onset, the acidosis is designated as an '
acute respiratory acidosis'. The body's initial compensatory response is limited during this phase.
As the body's renal compensatory response increases over the next few days, the pH returns towards the normal value and the condition is now a '
chronic respiratory acidosis'.
The differentiation between acute and chronic
is determined by time but occurs because of the renal compensatory response (which is slow).
Causes of Respiratory Acidosis:
The arterial pCO
2 is normally maintained at a level of about 40 mmHg by a balance between production of CO
2 by the body and its removal by alveolar ventilation. If the inspired gas contains no CO
2 then this relationship can be expressed by:
paCO2 is proportional to VCO2 / VA
where:
VCO2 is CO2 production by the body
VA is Alveolar ventilation
An increase in arterial pCO
2 can occur by one of three possible mechanisms:
- Presence of excess CO2 in the inspired gas
- Decreased alveolar ventilation
- Increased production of CO2 by the body
CO
2 gas can be added to the inspired gas or it may be present because of rebreathing : Anaesthetists are familiar with both these mechanisms. In these situations, hypercapnia can be induced even in the presence of normal alveolar ventilation and normal carbon dioxide production by the body.
An adult at rest produces about 200mls of CO
2 per minute