Showing posts with label Acid Base Physiology: Acid Base Learner Series. Show all posts
Showing posts with label Acid Base Physiology: Acid Base Learner Series. Show all posts
14.5.16
Regulation of Intracellular Hydrogen Ion Concentration
Importance of Intracellular [H+]
The most important [H+] for the body is the intracellular [H+]
Why? Because of its profound effects on metabolism and other cell processes which occur due to the effects of [H+] on the degree of ionisation of intracellular compounds. Specifically:
- Small molecule effect: Intracellular trapping function -due to the ionisation of metabolic intermediates.
- Large molecule effect: Effects on protein function: The function of many intracellular proteins (esp the activities of enzymes) is altered by effects on the ionisation of amino acid residues (esp histidine residues)
Renal Regulation of Acid-Base Balance
Renal Regulation of Acid-Base Balance
The organs involved in regulation of external acid-base balance are the lungs are the kidneys.
The lungs are important for excretion of carbon dioxide (the respiratory acid) and there is a huge amount of this to be excreted: at least 12,000 to 13,000 mmols/day.
In contrast the kidneys are responsible for excretion of the fixed acids and this is also a critical role even though the amounts involved (70-100 mmols/day) are much smaller. The main reason for this renal importance is because there is no other way to excrete these acids and it should be appreciated that the amounts involved are still very large when compared to the plasma [H+] of only 40 nanomoles/litre.
There is a second extremely important role that the kidneys play in acid-base balance, namely the reabsorption of the filtered bicarbonate. Bicarbonate is the predominant extracellular buffer against the fixed acids and it important that its plasma concentration should be defended against renal loss.
In acid-base balance, the kidney is responsible for 2 major activities:
- Reabsorption of filtered bicarbonate: 4,000 to 5,000 mmol/day
- Excretion of the fixed acids (acid anion and associated H+): about 1 mmol/kg/day.
Both these processes involve secretion of H+ into the lumen by the renal tubule cells but only the second leads to excretion of H+ from the body.
The renal mechanisms involved in acid-base balance can be difficult to understand so as a simplification we will consider the processes occurring in the kidney as involving 2 aspects:
- Proximal tubular mechanism
- Distal tubular mechanism
Respiratory Regulation of Acid-Base Balance: Acid Base Learner Series
How is the Respiratory System Linked to Acid-base Changes?
‘Respiratory regulation’ refers to changes in pH due to pCO2 changes from alterations in ventilation. This change in ventilation can occur rapidly with significant effects on pH. Carbon dioxide is lipid soluble and crosses cell membranes rapidly, so changes in pCO2 result in rapid changes in [H+] in all body fluid compartments.
A quantitative appreciation of respiratory regulation requires knowledge of two relationships which provide the connection between alveolar ventilation and pH via pCO2. These 2 relationships are:
- First equation - relates alveolar ventilation (VA) and pCO2
- Second equation - relates pCO2 and pH.
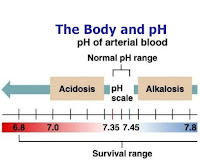
Acid Base Physiology: Acid Base Learner Series
Acid Base Physiology
Each day there is always a production of acid by the body’s metabolic processes and to maintain balance, these acids need to be excreted or metabolised. The various acids produced by the body are classified as respiratory (or volatile) acids and as metabolic (or fixed) acids. The body normally can respond very effectively to perturbations in acid or base production.
Respiratory Acid
The acid is more correctly carbonic acid (H2CO3) but the term 'respiratory acid' is usually used to mean carbon dioxide. But CO2 itself is not an acid in the Bronsted-Lowry system as it does not contain a hydrogen so cannot be a proton donor. However CO2 can instead be thought of as representing a potential to create an equivalent amount of carbonic acid. Carbon dioxide is the end-product of complete oxidation of carbohydrates and fatty acids. It is called a volatile acid meaning in this context it can be excreted via the lungs. Of necessity, considering the amounts involved there must be an efficient system to rapidly excrete CO2.
The amount of CO2 produced each day is huge compared to the amount of production of fixed acids. Basal CO2production is typically quoted at 12,000 to 13,000 mmols/day.
Increased levels of activity will increase oxygen consumption
Subscribe to:
Comments (Atom)