2.7.16
26.5.16
HIGHEST Yield Pearls for MRCP 1 2 & PACES
HIGHEST Yield Pearls for MRCP 1 2 & PACES
1) Hepatitis "D" Virus has HIGHEST mortality in ALL people EXCEPT#Pregnancy.
In Pregnant women: MOST LETHAL is Hepatitis E virus.
2) "While calculating Serum Osmolality the serum Na is multiplied by 2 to account for the accompanying ANIONS (mostly Cl- & HCO3-)."
3) Most Common cause of Metastasis to LIVER is primary cancers of Lung.
LLLLLungs send metastasis to LLLLLLiver.
4) Ketamine is the ONLY induction agent that causes Bronchodilation.
Again:
Ketamine is the ONLY induction agent that causes Bronchodilation.
5) AANNencephaly: failure of AANNterior neuropore to close.(at day 25)
SSSpinda bifida: failure of poSSSterior neuropore to close.(at day 27)
6) Muscle Spindles: Innervated by 1a fibers - cause skeletal muscle to CONTRACT.
GTOs: innervated by 1b - cause contracting muscle to RELAX
7) In SSSpontaneous pneumothorax: trachea shifts to ipSSSilateral side.
In TTTension pneumothorax: trachea deviates to conTTTralateral side.
8) Antibiotics SAFE in pregnancy:
All Penicillins.
All Cephalosporins.
All Carbapenems.
Aztreonam.
Azithromycin.
Nitrofurantoin (Avoid in last trimester bcoz can cause hemolysis in G6PD deficient fetus).
Metronidazole (SAFE in ALL trimesters).
1) Hepatitis "D" Virus has HIGHEST mortality in ALL people EXCEPT#Pregnancy.
In Pregnant women: MOST LETHAL is Hepatitis E virus.
2) "While calculating Serum Osmolality the serum Na is multiplied by 2 to account for the accompanying ANIONS (mostly Cl- & HCO3-)."
3) Most Common cause of Metastasis to LIVER is primary cancers of Lung.
LLLLLungs send metastasis to LLLLLLiver.
4) Ketamine is the ONLY induction agent that causes Bronchodilation.
Again:
Ketamine is the ONLY induction agent that causes Bronchodilation.
5) AANNencephaly: failure of AANNterior neuropore to close.(at day 25)
SSSpinda bifida: failure of poSSSterior neuropore to close.(at day 27)
6) Muscle Spindles: Innervated by 1a fibers - cause skeletal muscle to CONTRACT.
GTOs: innervated by 1b - cause contracting muscle to RELAX
7) In SSSpontaneous pneumothorax: trachea shifts to ipSSSilateral side.
In TTTension pneumothorax: trachea deviates to conTTTralateral side.
8) Antibiotics SAFE in pregnancy:
All Penicillins.
All Cephalosporins.
All Carbapenems.
Aztreonam.
Azithromycin.
Nitrofurantoin (Avoid in last trimester bcoz can cause hemolysis in G6PD deficient fetus).
Metronidazole (SAFE in ALL trimesters).
23.5.16
Free Exam Resources for doctors
Following are free resources for exam Practice
Specefically MRCP, PLAB, MRCGP and clinical revision
MRCPass
• Notes on topics covered in MRCP Part 1
• 130 MCQ questions
• Bookmark questions
• Choose questions by topic
• Track statistics
• Look back over wrong answers
Revise MRCP
• Covers MRCP Parts 1 and 2
• Over 5,000 MCQs arranged by topic
• Free to sign up
• Past papers going back to September 2010
Medexam.net
• Over 1,800 MCQs with detailed answers
Specefically MRCP, PLAB, MRCGP and clinical revision
MRCPass
• Notes on topics covered in MRCP Part 1
• 130 MCQ questions
• Bookmark questions
• Choose questions by topic
• Track statistics
• Look back over wrong answers
Revise MRCP
• Covers MRCP Parts 1 and 2
• Over 5,000 MCQs arranged by topic
• Free to sign up
• Past papers going back to September 2010
Medexam.net
• Over 1,800 MCQs with detailed answers
14.5.16
The Only MRCP Notes you will ever need 5th Edition: 2015 Edition
The Only MRCP Notes you will ever need 5th Edition
This is a solid book for consolidating your knowledge before the exam, Most of the data is updated than the 4th edition, But still few topics you should see through Passmedicine.com like ACS, STROKE, ARRHYTHMIA management.
Also latest Treatment for diabetes according to NICE December 2015 guidelines
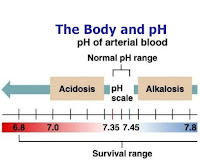
Regulation of Intracellular Hydrogen Ion Concentration
Importance of Intracellular [H+]
The most important [H+] for the body is the intracellular [H+]
Why? Because of its profound effects on metabolism and other cell processes which occur due to the effects of [H+] on the degree of ionisation of intracellular compounds. Specifically:
- Small molecule effect: Intracellular trapping function -due to the ionisation of metabolic intermediates.
- Large molecule effect: Effects on protein function: The function of many intracellular proteins (esp the activities of enzymes) is altered by effects on the ionisation of amino acid residues (esp histidine residues)
Renal Regulation of Acid-Base Balance
Renal Regulation of Acid-Base Balance
The organs involved in regulation of external acid-base balance are the lungs are the kidneys.
The lungs are important for excretion of carbon dioxide (the respiratory acid) and there is a huge amount of this to be excreted: at least 12,000 to 13,000 mmols/day.
In contrast the kidneys are responsible for excretion of the fixed acids and this is also a critical role even though the amounts involved (70-100 mmols/day) are much smaller. The main reason for this renal importance is because there is no other way to excrete these acids and it should be appreciated that the amounts involved are still very large when compared to the plasma [H+] of only 40 nanomoles/litre.
There is a second extremely important role that the kidneys play in acid-base balance, namely the reabsorption of the filtered bicarbonate. Bicarbonate is the predominant extracellular buffer against the fixed acids and it important that its plasma concentration should be defended against renal loss.
In acid-base balance, the kidney is responsible for 2 major activities:
- Reabsorption of filtered bicarbonate: 4,000 to 5,000 mmol/day
- Excretion of the fixed acids (acid anion and associated H+): about 1 mmol/kg/day.
Both these processes involve secretion of H+ into the lumen by the renal tubule cells but only the second leads to excretion of H+ from the body.
The renal mechanisms involved in acid-base balance can be difficult to understand so as a simplification we will consider the processes occurring in the kidney as involving 2 aspects:
- Proximal tubular mechanism
- Distal tubular mechanism
Respiratory Regulation of Acid-Base Balance: Acid Base Learner Series
How is the Respiratory System Linked to Acid-base Changes?
‘Respiratory regulation’ refers to changes in pH due to pCO2 changes from alterations in ventilation. This change in ventilation can occur rapidly with significant effects on pH. Carbon dioxide is lipid soluble and crosses cell membranes rapidly, so changes in pCO2 result in rapid changes in [H+] in all body fluid compartments.
A quantitative appreciation of respiratory regulation requires knowledge of two relationships which provide the connection between alveolar ventilation and pH via pCO2. These 2 relationships are:
- First equation - relates alveolar ventilation (VA) and pCO2
- Second equation - relates pCO2 and pH.
Acid Base Learner Series: Buffers
Definition of a Buffer
A buffer is a solution containing substances which have the ability to minimise changes in pH when an acid or base is added to it 1.
A buffer typically consists of a solution which contains a weak acid HA mixed with the salt of that acid & a strong base eg NaA. The principle is that the salt provides a reservoir of A- to replenish [A-] when A- is removed by reaction with H+.
Buffers in the Body
The body has a very large buffer capacity.
This can be illustrated by considering an old experiment (see below) where dilute hydrochloric acid was infused into a dog.
Acid Base Physiology: Acid Base Learner Series
Acid Base Physiology
Each day there is always a production of acid by the body’s metabolic processes and to maintain balance, these acids need to be excreted or metabolised. The various acids produced by the body are classified as respiratory (or volatile) acids and as metabolic (or fixed) acids. The body normally can respond very effectively to perturbations in acid or base production.
Respiratory Acid
The acid is more correctly carbonic acid (H2CO3) but the term 'respiratory acid' is usually used to mean carbon dioxide. But CO2 itself is not an acid in the Bronsted-Lowry system as it does not contain a hydrogen so cannot be a proton donor. However CO2 can instead be thought of as representing a potential to create an equivalent amount of carbonic acid. Carbon dioxide is the end-product of complete oxidation of carbohydrates and fatty acids. It is called a volatile acid meaning in this context it can be excreted via the lungs. Of necessity, considering the amounts involved there must be an efficient system to rapidly excrete CO2.
The amount of CO2 produced each day is huge compared to the amount of production of fixed acids. Basal CO2production is typically quoted at 12,000 to 13,000 mmols/day.
Increased levels of activity will increase oxygen consumption
Manchester Metropolitan Postgraduate Courses Fairs
Postgraduate Courses for 2016 entry
At the event you will be able to
attend subjects sessions, meet staff, pick up a prospectus and find out about
postgraduate loans.
The Faculty of Humanities, Languages
and Social Science is also pleased to announce we will be offering sixteen
scholarships to assist students with financing their Masters studies. Students
applying for full or part-time study on any taught Masters Programmes in the
Faculty are eligible for these scholarships which will pay 50% of the fees |
24.2.16
Acid Base Learner Series: Respiratory Acidosis in detail
A respiratory acidosis is a primary acid-base disorder in which arterial pCO2 rises to a level higher than expected.
At onset, the acidosis is designated as an 'acute respiratory acidosis'. The body's initial compensatory response is limited during this phase.
As the body's renal compensatory response increases over the next few days, the pH returns towards the normal value and the condition is now a 'chronic respiratory acidosis'.
The differentiation between acute and chronic
is determined by time but occurs because of the renal compensatory response (which is slow).
Causes of Respiratory Acidosis:
The arterial pCO2 is normally maintained at a level of about 40 mmHg by a balance between production of CO2 by the body and its removal by alveolar ventilation. If the inspired gas contains no CO2 then this relationship can be expressed by:
paCO2 is proportional to VCO2 / VA
where:
VCO2 is CO2 production by the body
VA is Alveolar ventilation
An increase in arterial pCO2 can occur by one of three possible mechanisms:VCO2 is CO2 production by the body
VA is Alveolar ventilation
- Presence of excess CO2 in the inspired gas
- Decreased alveolar ventilation
- Increased production of CO2 by the body
An adult at rest produces about 200mls of CO2 per minute
Acid Base Balance Series: What is the 'osmolar gap'?
NB: 'Osmolar gap' has several alternative names: 'osmol gap', 'osmole gap', 'osmolarity gap' & 'osmolal gap'; these all refer to the same thing. For consistency, the term "osmolar gap" is used exclusively through this book.
So osmolality is a measure of the number of particles present in a unit weight of solvent. It is independent of the size, shape or weight of the particles. It can only be measured by use of a property of the solution that is dependent on the particle concentration. These properties are collectively referred to as Colligative Properties. Osmolality is measured in the laboratory by machines called osmometers. The units of osmolality are mOsm/kg of solute
Osmolarity is calculated from a formula which represents the solutes which under ordinary circumstances contribute nearly all of the osmolality of the sample. There are many such formulae which have been used. One widely used formula for plasma which is used at my hospital is:
What is the 'osmolar gap'?
Osmolarity is calculated from a formula which represents the solutes which under ordinary circumstances contribute nearly all of the osmolality of the sample. There are many such formulae which have been used. One widely used formula for plasma which is used at my hospital is:
Calculated osmolarity = (1.86 x [Na+]) + [glucose] + [urea] + 9
Note regarding units: For the above equation, all concentrations are in mmol/l, and not mg/100mls. The result will then be in mOsm/l of solution. This equation is often expressed differently in North America where glucose & blood urea nitrogen (BUN) are reported inAcid Base Learner Series: The Urinary Anion Gap
The Urinary Anion Gap
The cations normally present in urine are Na+, K+, NH4+, Ca++ and Mg++.
The anions normally present are Cl-, HCO3-, sulphate, phosphate and some organic anions.
Only Na+, K+ and Cl- are commonly measured in urine so the other charged species are the unmeasured anions (UA) and cations (UC).
Because of the requirement for macroscopic electroneutrality, total anion charge always equals total cation charge, so:
The cations normally present in urine are Na+, K+, NH4+, Ca++ and Mg++.
The anions normally present are Cl-, HCO3-, sulphate, phosphate and some organic anions.
Only Na+, K+ and Cl- are commonly measured in urine so the other charged species are the unmeasured anions (UA) and cations (UC).
Because of the requirement for macroscopic electroneutrality, total anion charge always equals total cation charge, so:
Cl- + UA = Na+ + K+ + UC
Rearranging:Urinary Anion Gap = ( UA - UC ) = [Na+]+ [K+] - [Cl-]
Clinical Use
Key Fact: The urinary anion gap can help to differentiate between GIT and renal causes of a hyperchloraemic metabolic acidosis.
It has been found experimentally that the Urinary Anion Gap (UAG) provides a rough index of urinary ammonium excretion. Ammonium is positively charged so a rise in its urinary concentration (ie increased unmeasured cations) will cause a fall in UAGAcid Base Learner Series: The Delta Ratio
Definition
This Delta Ratio is sometimes useful in the assessment of metabolic acidosis. As this concept is related to the anion gap (AG) and buffering, it will be discussed here before a discussion of metabolic acidosis. The Delta Ratio is defined as:Delta ratio = (Increase in Anion Gap / Decrease in bicarbonate)
Others have used the delta gap (defined as rise in AG minus the fall in bicarbonate), but this uses the same information as the delta ratio and has does not offer any advantage over it.How is this useful?
In order to understand this, consider the following:If one molecule of metabolic acid (HA) is added to the ECF and dissociates, the one H+ released will react with one molecule of HCO3- to produce CO2 and H2O. This is the process of buffering. The net effect will be an increase in unmeasured anions by the one acid anion A- (ie anion gap increases by one) and a decrease in the bicarbonate by one.
Now, if all the acid dissociated in the ECF and all the buffering was by bicarbonate, then the increase in the AG should be equal to the decrease in bicarbonate so the
Subscribe to:
Posts (Atom)